-

What Is Gasification?

Gasification is a process that uses heat, pressure, and steam to convert materials directly into a gas. It has been in use for many years and involves breaking down complex organic molecules and carbon, in both liquid and solid state, into simple gases.
Gasification is a thermal process, but it is designed to produce a synthetic gas, or syngas, as the main product, rather than the flue gas produced by a similar thermal process, combustion (i.e., incineration). The syn-gas consists mainly of carbon dioxide and H2 and contains a large percentage of the inherent chemical energy of the input fuel.
Rather than treating waste in an oxygen-free environment, as is the case with pyrolysis, gasification occurs in the presence of a controlled amount of air, sufficient to allow only partial combustion of a small portion of the fuel input. The thermodynamics of the process are, therefore, significantly different from those of pyrolysis. Oxygen in the air facilitates the breakdown of heavy organic compounds in the waste, thereby optimizing the yield of syngas. Most of the gases produced are flammable and are, therefore, generally used as fuel in processes or applications where such gases are required. When mixed with air, syn-gas can be used in gasoline or diesel engines with minimal modifications to the engine.
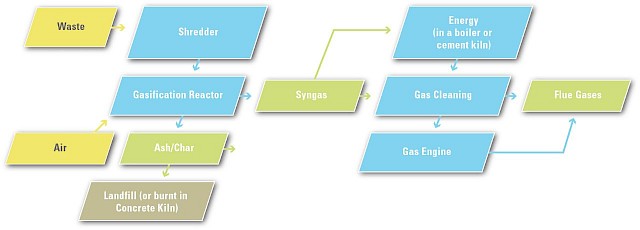
Our gasification technologies differ in several technical aspects but rely on four key engineering factors:
Typical raw materials used are coal, petroleum-based materials, and organic materials. The feedstock is prepared and fed, in either dry or slurried form, into a sealed reactor chamber called a gasifier, where it is subjected to high heat, pressure, and either an oxygen-rich or oxygen-starved environment. Most commercial gasification technologies do not use oxygen; however, all require an energy source to generate heat, which initiates the process.
We support our client’s through our quoting and project development by providing a wealth of information and specifications about Klean Industries systems and technologies.
©2026 All Rights Reserved. Terms of Use